Neat cutting oil and grinding oil – High-quality machining oil for cutting processes
Neat cutting oil and grinding oil, often also referred to as machining oil, is used, for example, for tapping, drilling or grinding and ensures optimum results with various materials. Our high-quality machining oils are based on the latest generation of oils. In the advanced gas-to-liquid (GTL) process, very pure, synthetic base oils are obtained from natural gas, which are free from organic nitrogen, heavy metals, zinc and chlorine compounds. The process safety of our GTL oils has been proven by various approvals from well-known customers in the automotive and supplier industry as well as from machine manufacturers. Discover our wide range of cutting oil and grinding oil.

What makes our cutting oil and grinding oil stand out?
Our high-performance machining oils - the Oemeta OEMETOL - are characterized by a very high flash point and a very low tendency to evaporate. This ensures good occupational safety and lower consumption. The higher lubricating performance and improved foaming behavior of GTL oils compared to conventional mineral or hydrocrack oils also reduce wear and improve process reliability.
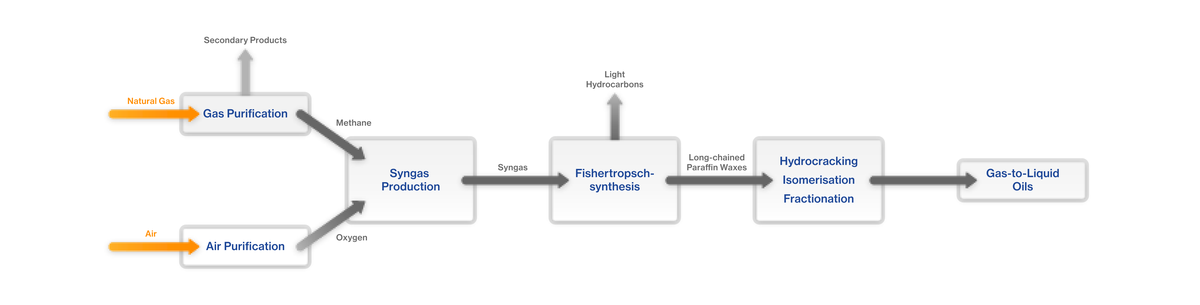
What is the GTL (Gas to Liquid) process?
The GTL process involves mixing natural gas and oxygen to form syngas. This is combined by means of a Fischer-Tropsch synthesis to form long-chained paraffin waxes. In the next step – the hydrocracking process – the long-chained paraffin waxes are converted into liquid hydrocarbons (saturated short-chained paraffins) by means of cracking, isomerisation and subsequent fractionation.

- Neutral odour
- Very low emission
- Low foaming and high air separation characteristics
- Improved filterability and greater economic efficiency due to low density
- Not cytotoxic – tested in accordance with EN ISO 10993-5:2009
(body compatibility/biocompatibility as a prerequisite in medical technology, e.g. for the use of implants) - Outstanding lubricating performance due to selected EP and AW additives
Wide selection of cutting oil and grinding oil
With our product finder, you can select the desired parameters and immediately receive the right machining oil for your individual requirements.
Can't find what you're looking for?
For individual inquiries or further information, please contact us at:
csrusa@![]() oemeta.com / +1 801 953 0134 / Contact form
oemeta.com / +1 801 953 0134 / Contact form